nature reviews gastroenterology & hepatology
FibroFind are delighted with the publishing of an article by Professor Derek Man, Chief Scientific Officer of FibroFind with the focus on encouraging a human-centric approach for the design and testing of antifibrotics in fatty liver disease, as part of the theme precision cut tissue slices (PCLS) were highlighted as an essential part of the process.
The link to the article in Nature Reviews Gastroenterology & Hepatology is below with the journal focused entirely on liver fibrosis, including other review articles from leaders in the field.
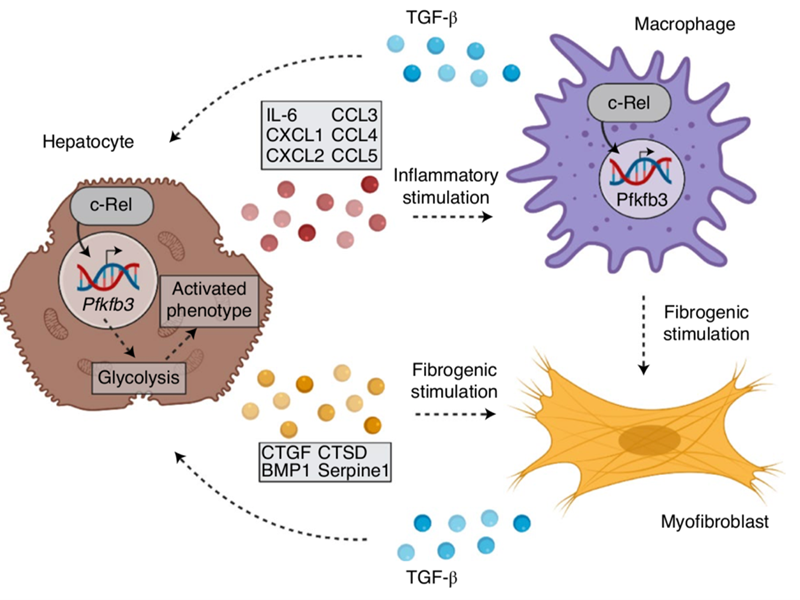
c-Rel orchestrates energy-dependent epithelial and macrophage reprogramming in fibrosis
Here FibroFind scientists collaborated in a study led by Professor Fiona Oakley (FibroFind founding scientists and Director) that describes how a factor (c-Rel) that is best known for its role in the immune system also controls metabolic processes that are important for fibrosis. The identified c-Rel/Pfkfb3 pathway is proposed as a target for the development of anti-fibrotic drugs.
The FibroFind precision-cut liver slice model and its proprietary tissue bioreactor technology was vital to this work.
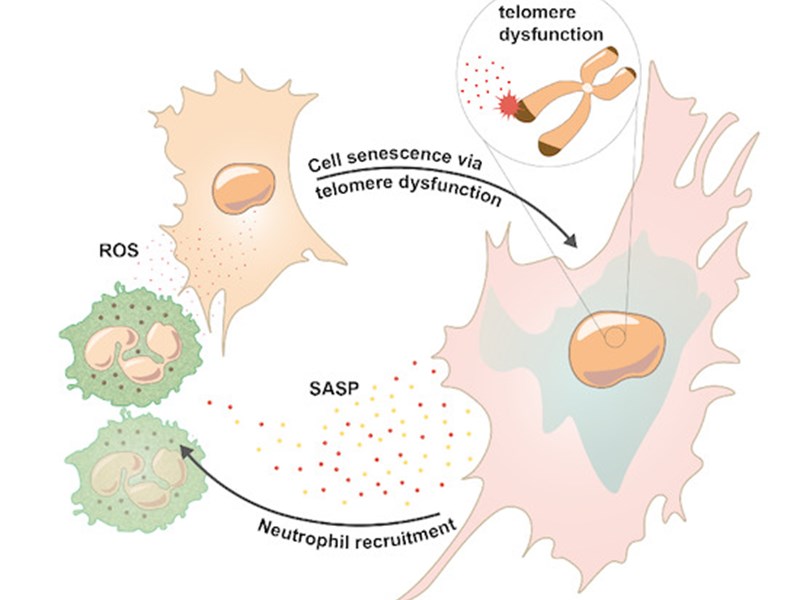
Neutrophils induce paracrine telomere dysfunction and senescence in ROS‐dependent manner
In this study which was co-led by FibroFind Chief Scientific Officer, Professor Derek Mann, the neutrophil which defends against infection can stimulate cellular senescence, this a condition in which cells adopt a pro-inflammatory state. As neutrophils accumulate in chronic diseases this discovery reveals a new mechanism by which inflammation is perpetuated and identifies the neutrophil as a target for therapeutic manipulation.
FibroFind’s proprietary tissue bioreactor technology was vital to this work in showing that neutrophils can cause hepatocytes within intact human liver to undergo senescence, this represents a major advance in being able to study senescence in an intact human tissue system.

A Bioreactor Technology for Modeling Fibrosis in Human and Rodent Precision-Cut Liver Slices
Fibrofind is able to keep human organ slices in an intact and active state for 7 days. We have optimised induction of fibrogenesis/fibrosis in normal PCS using a specific set of stimuli, which we are able to readily block using an inhibitor, this providing a positive control for any treatment that you may want us to test.
Our technology platform is detailed in our recently published paper in Hepatology journal that describes the PCLS (Liver) technology and associated outputs

Immunomodulatory Effects of Lenvatinib Plus Anti–Programmed Cell Death Protein 1 in Mice and Rationale for Patient Enrichment in Hepatocellular Carcinoma
Collaborative study with a top liver cancer team in Barcelona where we reported the ability of a cancer drug Lenvatinib to combine with immunotherapy to stimulate the immune system to suppress the growth of liver cancers. Our contribution was providing key data using a new in vivo experimental model for liver cancer developed in the NFRG.

Moderate Exercise Inhibits Age-Related Inflammation, Liver Steatosis, Senescence, and Tumorigenesis
Describes the beneficial effects of modest exercise on age-related liver disease and cancer. The paper also explains the mechanisms by which exercise can suppress liver inflammation, fat deposition, senescence and cancer. It was highlighted in the national news through a press release by CRUK - https://news.cancerresearchuk.org/2021/02/15/gentle-regular-exercise-could-reduce-the-risk-of-liver-cancer-in-mice/

CXCR2 inhibition enables NASH-HCC immunotherapy
Landmark paper that describes a new treatment for liver cancer that develops on the background of fatty liver disease and highlights the potential for neutrophils which are specialised immune cells to contribute to cancer immunotherapy. The work was the basis for obtaining for a multi-centre UK clinical trial (CUBIC) in advanced liver cancer that is now underway.

A Mammalian Target of Rapamycin- Perilipin 3 (mTORC1-Plin3) Pathway is essential to Activate Lipophagy and Protects Against Hepatosteatosis
Paper that describes a new molecular pathway mTORC1-Plin3) by which liver cells break down lipids to protect the liver from development of fatty liver disease which can progress to cirrhosis and/or cancer. Fibrofinds precision cut liver slice technology was an important feature of the experimental work in this paper and reported for the first time that fat metabolism can be studied in our liver slice model system.

TREM-2 defends the liver against hepatocellular carcinoma through multifactorial protective mechanisms
Collaborative piece of work with a team in Spain led by my former mentee Jesus Maria Banales, it reports how a molecule that she worked on while in Newcastle has an important role to play in protecting the liver against cancer development through multiple mechanisms.

The E3 ubiquitin ligase RNF115 regulates phagosome maturation and host response to bacterial infection
This paper is a collaboration with Matthias Trost at Newcastle University. – Provides new insights into how bacterial infections are cleared by macrophages (a specialist immune cell).

Claspin haploinsufficiency leads to defects in fertility, hyperplasia and an increased oncogenic potential
This paper is a collaboration with Neil Perkins and Jill Hunter at Newcastle University.

A neutrophil–B-cell axis impacts tissue damage control in a mouse model of intraabdominal bacterial infection via Cxcr4
This paper is a collaboration with the group of Sylvia Knapp in Vienna.

c- Rel– dependent Chk2 signaling regulates the DNA damage response limiting hepatocarcinogenesis
This paper describes a new molecular pathway where the transcription factor c-Rel regulates the cellular repair response of hepatocytes to genotoxic injury which is important to prevent the development of liver cancer.

Role of Hepatocyte Senescence in the Activation of Hepatic Stellate Cells and Liver Fibrosis Progression
This is a collaboration with Dominic Aravinthan in Nottingham University showing that senescent hepatocytes secrete factors that can promote scar cell formation in the liver and promote liver fibrosis progression.

Copyright © 2021 JoVE Creative Commons Attribution 3.0 License jove.com September 2021 • 175 • e62868 • Page 1 of 15 A Three-Dimensional Spheroid Model to Investigate the Tumor-Stromal Interaction in Hepatocellular Carcinoma
Describes a methodology to make liver cancer cell spheroids and test therapies.

Sulfatase-2 from Cancer Associated Fibroblasts: An Environmental Target for Hepatocellular Carcinoma?
Provides new insights into the biology of liver cancer and describes how Sulfatase 2 produced in cancer associated fibroblasts promotes lliver cancer growth and resistance to therapy.

Key features of the environment promoting liver cancer in the absence of cirrhosis
Describes a new mouse model of liver cancer.

Suppression of insulin-induced gene 1 (INSIG1) function promotes hepatic lipid remodelling and restrains NASH progression
Collaboration with Toni Vidal-Puig in Cambridge. Provides new insights in to the mechanisms of NAFLD pathogenesis and revealed that SCAP/SREBP/INSIG1 trio governs transcriptional programmes that protect the liver from lipotoxic insults in NASH.
Non-invasive synchronous monitoring of neutrophil migration using whole body near-infrared fluorescence-based imaging
A method to track two cell types in vivo using spectral unmixing.

Anti–Cholestatic Therapy with Obeticholic Acid Improves Short-Term Memory in Bile Duct–Ligated Mice
This paper showed that obeticholic acid (OCA), a farnesoid X receptor agonist and second-line anti-cholestatic agent for treating PBC improved cognitive function, hippocampal connectivity and reduced neuronal senescence in a mouse model of cholestatic liver disease where cognitive impairment is a feature of the model. To extend the observations to a human system, cultured human neurons (stem cell derived) with cholestatic serum induced senescence, and, this was reversed by OCA.
